CHEM0039 2021 SAMPLE PAPER
Hello, dear friend, you can consult us at any time if you have any questions, add WeChat: daixieit
CHEM0039 2021 SAMPLE PAPER
Candidates should attempt ALL questions. Each question is marked out of 25 and the numbers in square brackets in the right-hand margin indicate the provisional allocation of marks to the subsections of a question.
1. Answer ALL parts
(a) (i) Describe the operation of a Solid Oxide Fuel Cell (SOFC), including
which materials are used as electrodes and electrolyte, operating temperature, which fuel can be used, the typical reactions taking place
at the electrodes.
(ii) Si and Sn are two materials of interest as alternative anodes for Li-ion
batteries. Discuss their benefits and limitations relative to graphite.
(iii) Olivine-structured LixFePO4 can operate as intercalation cathode, with x varying between 0.0 and 1.0. Calculate the specific capacity of FePO4; its specific energy, assuming that the average cell voltage is 3.5V, and its specific power when the battery operates at C=0.2
b) (i) Lithium-air batteries and conversion reactions are two of the research directions explored to improve upon the current energy density of Li- ion batteries (LIBs). Discuss the principles upon which Li-air and conversion electrodes are based, and why their theoretical energy density is higher than that of current LIBs.
(ii) Describe how oxide-ion conductivity in ZrO2 can be enhanced by doping. Write the corresponding reactions using the Kröger-Vink notation.
2. Answer ALL parts.
(a) You are asked to provide a recommendation for a thin film deposition system.
The requirements from the customer are:
Deposition of films on flat glass substrates as part of a continuous manufacturing process
Low cost
Ability to deposit a variety of chemical compositions
Recommend a thin film deposition technique, and explain how it can best match
each ofthese requirements.
(b) You are asked by a world leading university to establish a thin film research
facility. The university wishes to research superlattices, and is interested in the relationship between the structure of superlattice films and their properties. The university is considering CVD, ALD, PVD and MBE as possible deposition techniques.
(i) Stating your reasons, recommend ONE of these techniques to the university.
(ii) Describe what analysis techniques the university will need in order to properly characterise the structure of the films.
(iii) Pick ONE of the techniques you mentioned in part (ii). Discuss the information that can be obtained from that analysis technique. Describe
any limitations ofthe technique.
3. Answer ALL parts
(a) (i) Define polymorphism and explain how it can affect drug formulation.
(ii) What do you understand by the following terms: crystal form, crystal morphology and supramolecular synthon?
(iii) Describe the difference between a cocrystal and a solid solution. What are the characteristics of a pair of molecules forming a cocrystal,
|
compared to that ofmolecules that form a solid solution? |
(iv) Define a halogen bond and explain how it forms.
(v) What are the geometrical requirements for olefins to undergo a [2+2] photodimerisation in the solid state.
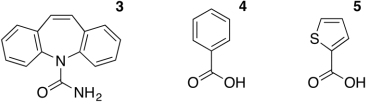
(b) (i) The figure below shows the molecular structures of carbamazepine (3), 2-thiophenecarboxylic acid (4) and benzoic acid (5).
|
|
The powder patterns shown below relate to solids that were obtained by milling:
E: carbamazepine and 2-thiophenecarboxylic acid in a 1:1 ratio
F: carbamazepine and benzoic acid in a 1:1 ratio
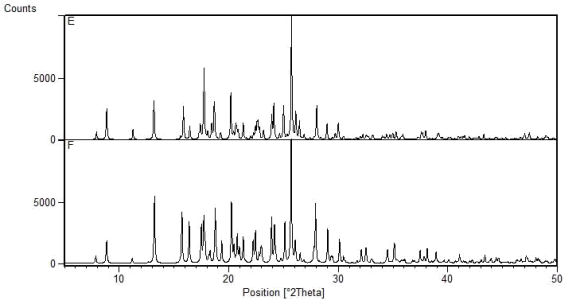
Comment on how the crystal structures of the two solids are related.
Explain why patterns E and F are so similar?
(ii) Figures a) and b) show structural features of a 1:1:1 cocrystal hydrate composed of caffeine and paracetamol. Explain why this cocrystal has the potential to perform better as an active ingredient in a tablet than a
physical mixture of caffeine and paracetamol.

2022-01-11