CHE00016I Core 4a: Molecules in Action 2019-20
Hello, dear friend, you can consult us at any time if you have any questions, add WeChat: daixieit
CHE00016I
BSc Chemistry and MChem Degree Examinations 2019-20
Chemistry
Core 4a: Molecules in Action
Section A
Answer Question 1
Question 1 - answer each question in a separate booklet
(a) Identify and briefly discuss four characteristics which have enabled nature to select (4)
carbon, oxygen, nitrogen and hydrogen as the major components of biological molecules such as proteins and nucleic acids.
(b) Describe the oil drop model of folded globular proteins in aqueous solution. Identify (4)
the thermodynamic basis for the stabilisation ofthe fold and describe the interactions involved.
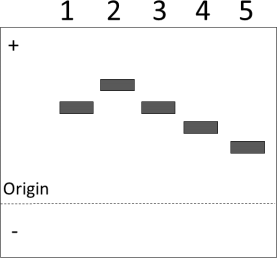
(c) The figure below shows the results of a gel electrophoresis experiment run at pH 7. It compares a wild type protein (Lane 1) with four variant proteins with amino acid residue substitutions.
Giving your reasoning in each case, identify the lanes associated with proteins (4)
containing the following substitutions:
● phenylalanine to aspartic acid;
● phenylalanine to lysine;
● aspartic acid to lysine.
Note: amino acid structures are given in the databook.
(d) The sequence below is of a segment ofmessenger RNA that includes a STOP codon at the end ofthe coding sequence of a monomeric protein X.
5’ ACG GAA CUG CCG UUC GGG UGA CCC UCA 3’
(i) Identify the last five residues of the protein. (2) Note: the genetic code is given in the databook.
(ii) Write down the sequence ofthe template DNA strand from which this (2)
messenger RNA was transcribed.
(e) A mutant form of protein X that differs only by a single amino acid residue substitution (4)
forms dimers under oxidative conditions. Sequence analysis shows that the messenger RNA sequence is altered to:
5’ ACG GAA CUG CCG UGC GGG UGA CCC UCA 3’
Identify the change in the protein sequence and suggest how the protein forms dimers.
![]()
![]()
![]()
Section B
Answer two questions from this section
Question 2 - answer each question in a separate booklet
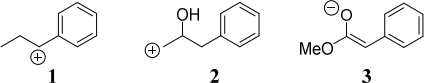
(a) Give synthetic equivalents for each ofthe synthons 1, 2 and 3. (3)
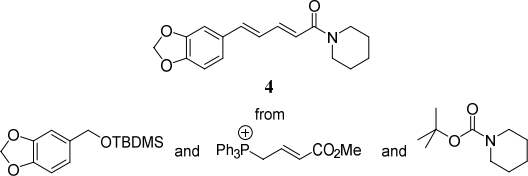
(b) Devise a synthetic route to piperine 4, a component of black pepper, from the starting (8)
materials shown below. Your answer should show the retrosynthetic analysis and the forward synthesis, indicating suitable reagents, and identifying any steps that are stereoselective.
Note: more than one synthetic step is required.![]()
![]()
![]()
(c) The synthesis of substituted benzene A is shown below.![]()
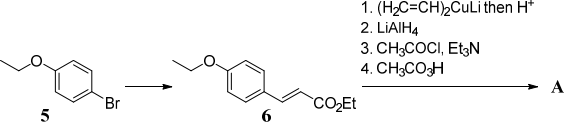
(i) Suggest reagents for converting phenol (PhOH) into 5 in 2-steps and explain (3) how your approach gives the correct regioselectivity.
(ii) Suggest reagents for converting 5 into 6 using a Heck reaction and account (2)
for the regioselectivity.
(iii) Give the structure ofA and explain how it is formed from 6. Account for the (4)
regioselectivity in Step 1.
![]()
![]()
Question 3 - answer each question in a separate booklet
(a) Compound 4 can be synthesised from dimethyl hexanedioate, 1.
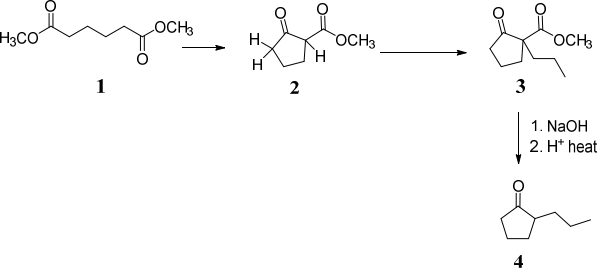
![]()
(i) Give suitable reagents for the conversion of 1 into 2 and draw a curly arrow (3) mechanism for the reaction.
(ii) Give suitable reagents for the conversion of 2 into 3, explain your choice of (4)
reagents and show a curly arrow mechanism for the conversion.
(iii) Give a curly arrow mechanisms for the conversion of3 into 4. (2)
Note: more than one step is required.
(b) Ketone 4 can be methylated to give either ketone 5 or ketone 6.
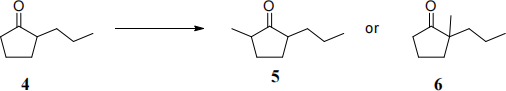
For both reactions give suitable reagents and draw curly arrow mechanisms. (6)
(c) Give a full retrosynthetic analysis to show how 7 can be synthesised from 8, 9 and 10. (5) Using your retrosynthesis, show a forward synthesis giving suitable reagents for each step.
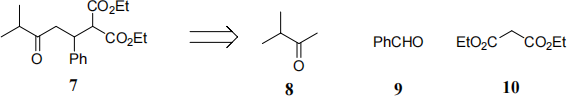
Question 4 - answer each question in a separate booklet
(a) (i) Estimate the pressure difference between the surface and the bottom of a body (2)
of water having a depth of 50 m.
Note: assume values of 1 kg dm![]() 3 for the density ofwater and 10 m s
3 for the density ofwater and 10 m s![]() 2 for gravitational acceleration.
2 for gravitational acceleration.
(ii) Using the Henry’s law constant kH = 2.58 × 108 Pa, calculate the mole-fraction (3)
solubilities of CO2 at:
![]() atmospheric pressure (105 Pa);
atmospheric pressure (105 Pa);
![]() the pressure you calculated in part (a)(i).
the pressure you calculated in part (a)(i).
(b) A lake in Africa suddenly released CO2, a phenomenon termed limnic eruption, when the
bottom lake waters were destabilised and ascended to the surface. The area ofthe lake was 1.5 km2 and its depth 100 m.
(i) The difference in mole-fraction solubility of CO2 between the surface and (4) bottom ofthe lake was 3.8 × 10![]() 3 . Assuming that the density ofwater is not
3 . Assuming that the density ofwater is not
affected by pressure, calculate the moles of CO2 released from 1 m3 ofwater when it transferred from the bottom to the surface ofthe lake.
(ii) Assume the lake to be ofuniform depth. Using your answer to part (b)(i) (3)
calculate, in moles, the total amount of CO2 released from the bottom 1 m layer ofwater in the lake when it was transferred to the surface ofthe lake. Hence, calculate the volume ofthe CO2 released under atmospheric pressure at 300 K.
(c) A propane tank, when exposed to fire, poses a serious risk of explosion.
(i) Propane requires a pressure of 1220 kPa to keep it liquid at 37.8 oC. Using the (3) enthalpy of vaporization ofpropane, 24.5 kJ mol![]() 1, calculate the pressure
1, calculate the pressure
required at 100 oC under liquid-vapour equilibrium.
(ii) A pressure reliefvalve on the tank is designed to release the gas when the (3)
internal pressure reaches 1700 k Pa. Assuming liquid-vapour equilibrium, calculate the temperature at which the valve opens.
(iii) Giving your reasons, briefly discuss whether the temperature-dependence of (2)
vapour pressure is steeper for butane or for propane.
2021-12-29